
Chapter 3: The Chemical Basis for Life
Lesson 3.1: Unique Properties of Water
The glass in the picture above appears to be floating on water. How
can a solid float on water? Actually, nothing is floating on the water; rather
this picture depicts some of the most unique properties of water. What
appears to be a glass floating on water is really water splashing upward in
response to an item being dropped into the water. Water’s unique properties
of adhesion, surface tension, and its viscosity are what contribute to this
illusionary picture. These properties and others will be discussed in this
chapter.
Lesson Objectives
• Describe the distribution of Earth’s water.
• Identify water’s unique properties that support life on Earth.
• Define acids, bases, and pH.
• Explain why water is essential for life.
Vocabulary
● acids
● adhesion
● anabolic reaction
● bases
● boiling point
● buffer
● capillary action
● catabolic reaction
● cohesion
● freezing point
● hydrogen bond
● ion
● metabolism
● neutralization reaction
● pH
● polarity
● solubility
● solute
● solution
● solvent
● specific heat
● surface tension
● temperature
INTRODUCTION
Water, like carbon, has a special role in living things. It is needed by all known forms of life. All
organisms on Earth are made up mostly of water, thus water is the biological cornerstone of life. Water
is a simple molecule, containing just three atoms, two hydrogen and one oxygen. Nonetheless, water’s
structure gives it unique properties that help explain why it is vital to all living organisms. In fact, without
water, life would not be possible. This simple fact is why scientists are constantly looking for water on
other planets - the presence of water could indicate the presence of life.
WATER, WATER EVERYWHERE
Water is a common chemical substance on planet Earth. In fact, Earth is sometimes called the
‘‘water planet” because almost 75% of its surface is covered with water. If you look at Figure 3.1, you
will see where Earth’s water is found. The term water generally refers to its liquid state, and water is a
liquid over a wide range of temperatures and pressures on Earth. However, water also occurs on Earth
as a solid (ice) and as a gas (water vapor).
Chapter 3: Unique Properties of Water 57

Where Is All the Water?
Of all the water on Earth, about two
percent is stored underground in spaces between
rocks. A fraction of a percent exists in the air as
water vapor, clouds, or precipitation. Another
fraction of a percent occurs in the bodies of plants
and animals. So where is most of Earth’s water?
It’s on the surface of the planet. In fact, water
covers about 72 percent of Earth’s surface. Of
water on Earth’s surface, 97 percent is salt water,
found mainly in the ocean. Only 3 percent is
freshwater, which mostly found frozen in glaciers
and polar ice caps. The remaining fresh water
occurs in rivers, lakes, and other fresh water
features.
Although clean freshwater is essential to
human life, in many parts of the world it is in short
supply. The amount of fresh water is not the issue.
There is plenty of fresh water to go around,
because water constantly recycles on Earth.
However, fresh water is not necessarily located
where it is needed, and clean fresh water is not always available.
How Water Recycles
Like other matter on Earth, water is continuously recycled. Individual water molecules are
always going through the water cycle, in which molecules of water cycle through both the living and
nonliving parts of the biosphere. In fact, water molecules on Earth have been moving through the water
cycle for billions of years. In this cycle, water evaporates from Earth’s surface (or escapes from the
surface in other ways), forms clouds, and falls back to the surface as precipitation. This cycle keeps
repeating. Several processes change water from one state to another during the water cycle. They
include:
● Evaporation—Liquid water on Earth’s surface changes into water vapor in the atmosphere.
● Sublimation—Snow or ice on Earth’s surface changes directly into water vapor in the
atmosphere.
● Transpiration—Plants give off liquid water, most of which evaporates into the atmosphere.
● Condensation—Water vapor in the atmosphere changes to liquid water droplets, forming clouds
or fog.
● Precipitation—Water droplets in clouds are pulled to Earth’s surface by gravity, forming rain,
snow, or other type of falling moisture.
WATER THE MOST IMPORTANT MOLECULE FOR LIFE
Some may argue that DNA or proteins are the most important molecules for life. But many
would argue that water is really the most important molecule for life on Earth. What makes water so
important? Its unique chemical properties and structure allow water to be a polar molecule; which
Chapter 3: Unique Properties of Water 58

allows it to interact with many other molecules necessary for life. Most of the substances in a cell are
floating around in a water-based cytoplasmic environment.
STRUCTURE AND PROPERTIES OF WATER
You are probably already familiar with many of water’s properties.
For example, you know that water is tasteless, odorless, and transparent.
In small quantities, it is also colorless. However, when a large amount of
water is observed, as in a lake or the ocean, it is actually light blue in color.
The blue hue of water is caused by selective absorption and scattering of
white light. Transparency allows sunlight to pass through it. Sunlight is
needed by water plants and other water organisms for photosynthesis.
These properties of water are caused by its unique chemical structure.
Polarity
To understand some of water’s properties, you need to know
more about its chemical structure. Each molecule of water
consists of one atom of oxygen and two atoms of hydrogen, giving
it the chemical formula H
2
O. In each water molecule, the nucleus
of the oxygen atom (8 positively charged protons) attracts
electrons (e
-
) stronger than do the hydrogen nuclei (only one
positively charged proton). Because oxygen is more
electronegative —electron-greedy— than hydrogen, the O atom
hogs electrons and keeps them away from the H atoms. This gives
the oxygen end of the water molecule a partial negative charge,
while the hydrogen end has a partial positive charge. A difference
in electrical charge between different parts of a molecule is called
polarity. A polar molecule is is formed when part of the molecule
is positively charged and part of the molecule is negatively
charged.
Water is a polar molecule, with an unequal
distribution of charge throughout the molecule. The model in
Figure 3.3 shows the arrangement of oxygen and hydrogen
atoms in a water molecule. A water molecule has a bent or
angular (non-linear) shape, with an angle of about 105°.
When it comes to charged particles we say “opposites
attract”. In the case of water, the positive (hydrogen) end of
one water molecule is attracted to the negative (oxygen) end
of a nearby water molecule. These attractions are an example
of hydrogen bonds, weak interactions that form between a
hydrogen with a partial positive charge and a more
electronegative atom, such as oxygen. The hydrogen atoms
involved in hydrogen bonding must be attached to
electronegative atoms, such as O, N, or F. Nonetheless, they
are strong enough to hold together nearby molecules.
Hydrogen bonds can also form within a single large
organic molecule. For example, hydrogen bonds that form in
Chapter 3: Unique Properties of Water 59

a protein molecule bend the molecule into a distinctive shape, which is important for the protein’s
functions. Hydrogen bonds also hold together the two nucleotide chains of a DNA molecule.
Solvency
Water is one of the most common ingredients in solutions. A solution is a homogeneous mixture
composed of two or more substances. In a solution, one substance is dissolved in another substance,
forming a mixture that has the same proportion of substances throughout. The dissolved substance in a
solution is called the solute. The substance in which it is dissolved is called the solvent. An example of a
solution in which water is the solvent is salt water (refer back to Figure 3.4). In this solution, a
solid—sodium chloride (NaCl)—is the solute.
The ability of a solute to dissolve in a particular solvent is called solubility. Many chemical
substances are soluble in water. In fact, so many substances are soluble in water that water is called the
universal solvent. Water is a strongly polar solvent, and polar solvents are better at dissolving polar
solutes. Many organic compounds and other important biochemicals are polar, so they dissolve well in
water. On the other hand, strongly polar solvents like water cannot dissolve strongly nonpolar solutes
like oil. Did you ever try to mix oil and water? Even after being well shaken, the two substances quickly
separate into distinct layers.
Water is the solvent in solutions called acids and bases, which will be discussed later in this
chapter. To understand acids and bases, it is important to know more about pure water, in which
nothing is dissolved. In pure water (such as distilled water), a tiny fraction of water molecules naturally
breaks down, or dissociates, to form ions. An ion is an electrically charged atom or molecule.
The dissociation of pure water into ions is represented by the chemical equation:
2 H
2
O → H
3
O
+
+ OH
-
The products of this reaction are a hydronium ion (H
3
O+) and a hydroxide
ion (OH-). The hydroxide ion is negatively charged. It forms when a water
molecule donates, or gives up, a positively charged hydrogen ion. The hydronium
ion, modeled in Figure 3.5, is positively charged. It forms when a water molecule
accepts a positively charged hydrogen ion (H+).
Cohesion
Hydrogen bonds between water molecules explain some of water’s other properties. For
example, hydrogen bonds explain why water molecules tend to stick together. Did you ever watch water
drip from a leaky faucet or from a melting icicle? If you did, then you know that water always falls in
drops rather than as separate molecules. Cohesion
refers to the attraction of molecules for other
molecules of the same kind, and water molecules
have strong cohesive forces thanks to their ability
to form hydrogen bonds with one another.
Figure 3.6 Droplets of Dew. Drops of dew cling to a
blade of grass in this picture. Can you think of other
Chapter 3: Unique Properties of Water 60

examples of water forming drops? (Hint: What happens when rain falls on a newly waxed car?)
Surface Tension
Cohesive forces are responsible for surface tension, the tendency of a liquid’s surface to resist rupture
when placed under tension or stress. Water molecules at the surface (at the water-air interface) will
form hydrogen bonds with their neighbors, just like water molecules deeper within the liquid. However,
because they are exposed to air on one side, they will have fewer neighboring water molecules to bond
with, and will form stronger bonds with the neighbors they do
have. This results in a “skin” of water at the surface in which the
molecules are held together very tightly. Surface tension is a
measurement of the amount of force required to break this skin
on the surface of water. Other liquids have a surface tension as
well, but the surface tension in water is quite strong due to the
hydrogen bonds. You can view the “Jesus Christ” lizard, or
Basilisk lizard (Basiliscus basiliscus), found in Central and South
American rainforests) and how it uses surface tension to its
advantage through the following link:
Figure 3.7 Surface tension
allows water striders to
walk or skim over water
without sinking into it and
Adhesion
Another important physical property of water, is
adhesion. In terms of water, adhesion is the bonding of a water
molecule to another substance, such as water molecules bonding
to glass like the drops of water. This is an example of
intermolecular forces bonding. This process happens because
hydrogen bonds are special in that they break and reform with
great frequency. This constant rearranging of hydrogen bonds
allows a percentage of all the molecules in a given sample to
bond to another substance.
Chapter 3: Unique Properties of Water 61

Capillary Action
The grip-like characteristic that results from the water molecules property of adhesion helps to form
capillary action. Capillary action is the ability of a liquid to flow against gravity in a narrow space. An
example of capillary action is when you place a straw into a glass of water. The water seems to climb up
the straw before you even place your mouth on the straw. The water has created hydrogen bonds with
the surface of the straw, causing the water to adhere to the sides of the straw. As the hydrogen bonds
keep interchanging with the straw's surface, the water molecules interchange positions and some begin
to ascend the straw. A graphic description of this is explained .
• Capillary action occurs when water climbs
upward through a small space, defying gravity due
to the forces of adhesion and surface tension. The
image shows one example of capillary action - a
narrow straw was placed in a cup of water and the
water crept upwards through the straw.
• What causes the movement of water during
capillary action? The first factor is adhesion, the
attraction between water and another object. In
this case, adhesion attracted the water within the
straw to the surface of the straw. Molecules of
water which came in contact with the straw
tended to move upward along the inside of the
straw.
• Water's surface tension is so strong that, as
water is pulled upward along the straw's walls, the
water in between tends to be pulled upward also.
The downward pull of gravity prevents the central
water from rising quite as high as the water which
is adhered to the straw, so the result is a
meniscus, as shown in a graduated cylinder below.
Capillary action is important in moving water upwards through small spaces. Plants depend on
capillary action to move water upward from the roots to the leaves. In the soil, capillary action also
tends to move water upward between the soil particles. Water is moved through small blood vessels in
animals through capillary action also.
Chapter 3: Unique Properties of Water 62

TEMPERATURE MODERATION
In addition to the properties already discussed in this chapter, water also has a high heat
capacity. What this means is that water can absorb or release large amounts of energy in the form of
heat while only slightly changing its temperature. Energy must be absorbed to break hydrogen bonds,
and energy is released as heat when hydrogen bonds form. The energy that water initially absorbs
breaks hydrogen bonds between molecules. Only after these hydrogen bonds are broken does the
energy begin to increase the motion of the water molecules, which raises the temperature of the water.
When the temperature of water drops, hydrogen bonds reform, which releases a large amount of
energy in the form of heat. Temperature is a measure of the average kinetic energy (energy of motion)
of particles in a sample of matter. This physical property can determine the rate and extent to which
chemical reactions can occur within living systems. It is commonly measured in degrees Celsius (°C) or
Fahrenheit (°F).
Furthermore, water has a high specific heat, meaning it takes a lot of energy to raise or lower
the temperature of water. Specific heat is a measure of how much energy it takes to raise the
temperature of a substance. Specific heat (measured in cal/(g°C)) is a property that is unique to a given
type of matter. That’s why it’s called specific. Every substance has its own specific heat capacity, with
the specific heat capacity of water being 1 cal/(g°C). The specific heat capacity of water is much higher
than that of other common substances. For the sake of comparison, the specific heat capacity of oil is
about 0.5 cal/(g°C) and the specific heat capacity of aluminum is about 0.2 cal/(g°C). This means it takes
more heat to raise the temperature of water than it would take to raise the temperature of oil or
aluminum.
As a result, water plays a very important role in temperature regulation. Since cells are made up
of water, this property helps to maintain homeostasis. For example, since it takes such a large amount of
energy to change the state of water, sweating is a very effective method of cooling the body. In order to
evaporate, the sweat requires the input of a great deal of heat energy, some of which comes from our
bodies. So, as our sweat evaporates, we begin to feel cooler.
Freezing Point of Water
The freezing point, the temperature at which a liquid changes state to a solid, of water is 0°C
(32°F). Below this temperature, water is a solid (ice). Unlike most chemical substances, water in a solid
state has a lower density than water in a liquid state. This is because water expands when it freezes.
Again, hydrogen bonding is the reason. Hydrogen bonds cause water molecules to line up less efficiently
in ice than in liquid water. As a result, water molecules are spaced farther apart in ice, giving ice a lower
density than liquid water. A substance with lower density floats on a substance with higher density. This
explains why ice floats on liquid water, whereas many other solids sink to the bottom of liquid water.
In a large body of water, such as a lake or the ocean, the water with the greatest density always
sinks to the bottom. Water is most dense at about 4°C (39.2°F). As a result, the water at the bottom of a
lake or the ocean usually has temperature of about 4°C. In climates with cold winters, this layer of 4°C
water insulates the bottom of a lake from freezing temperatures. Lake organisms such as fish can
survive the winter by staying in this cold, but unfrozen, water at the bottom of the lake.
Boiling Point of Water
Boiling point is the temperature at which a liquid changes state into a gas. Hydrogen bonds,
which tend to hold water molecules together cause water to have a relatively high boiling point of 100°C
(212°F). This high boiling point means that liquid water turns into water vapor at a higher temperature
than would be expected due to the size and mass of the molecule. Substances similar to water have a
much lower boiling point because they lack hydrogen bonds. Most water on Earth is in a liquid state
rather than a gaseous state because of its high boiling point. Water in its liquid state is needed by all
Chapter 3: Unique Properties of Water 63

living things. Therefore, the availability of liquid water enables life to survive over much of the planet.
WATER AND LIFE
Water is essential to important chemical reactions in organisms. As a result, virtually all life
processes depend on water. Life as we know it could not exist without water. All life on Earth began in
water and evolved there for 3 billion years before spreading onto land. All present-day living organisms,
terrestrial and aquatic, remain tied to water.. For example, human beings can survive without food for a
few weeks, but only a week without water. Molecules of water are involved in many chemical reactions
necessary to sustain life. Most cells are surrounded by water, and three fourths of the Earth is
submerged in water.
Humans are composed of about 60-70 percent water (minus body fat). Water is crucial for
normal functioning of the body. Water’s ability to dissolve most biologically significant compounds
—from inorganic salts to large organic molecules— makes it a vital solvent inside organisms and cells.
Water is an essential part of most metabolic processes within organisms. Metabolism is the sum
of all body reactions, including those that build up molecules and /or store energy (anabolic reactions)
and those that break down molecules releasing energy (catabolic reactions). In anabolic reactions, water
is generally removed from small molecules in order to make larger molecules. In catabolic reactions,
water is used to break bonds in larger molecules in order to make smaller molecules.
Water is central to two related, fundamental metabolic reactions in organisms: photosynthesis
and cellular respiration. All organisms depend directly or indirectly on these two reactions. In
photosynthesis, cells metabolically use the energy in sunlight to change water and carbon dioxide into
glucose (C
6
H
12
O
6
) and oxygen (O
2
).
Anabolic reaction, represented by the chemical equation:
6 CO
2
+ 6 H
2
O + energy → C
6
H
12
O
6
+ 6 O
2
.
In cellular respiration, cells transfer chemical energy from glucose (by breaking it down) to ATP
(a usable energy-rich molecule) in the presence of oxygen and release energy, water, and carbon
dioxide.
Catabolic reaction, represented by the chemical equation:
C
6
H
12
O
6
+ 6 O
2
→ 6 CO
2
+ 6 H
2
O + energy
WATER ACTS AS A BUFFER
What is a buffer? Let's start with some background info first. To understand buffers, you need
to understand acids and bases. Acids are substances that release hydrogen ions (H+) into solution. For
example HCl, or hydrochloric acid, is a compound formed by ionic bonds. When you drop it in water, the
H+ and Cl- come apart, because as we said before, water is polar and will attack charged ions. As a
result, H+ ions are released into solution, which dramatically increases the concentration of H+. An
increase in the concentration of H+ causes an increase in acidity.
Bases, on the other hand, are substances that will bind to the free hydrogen ions (H+) that might
be floating around in solution. NaOH is an example of a base. Bases are also known as alkaline. When
you drop NaOH in water, the Na+ ions become separated from the hydroxide ions (OH-). Even though
the oxygen and hydrogen of OH- are bound together covalently, they still count as an ion because, as a
unit, they possess an extra electron, and therefore, have a net negative charge. When a stray OH- ion
encounters a free H+ ion, the ions bind. This results in the concentration of free H+ ions in solution to
decrease, which increases the basicity. Bases have more OH- ions than acids.
To summarize, acids release H+ ions into solution, and bases absorb them. Acids and bases are
Chapter 3: Unique Properties of Water 64
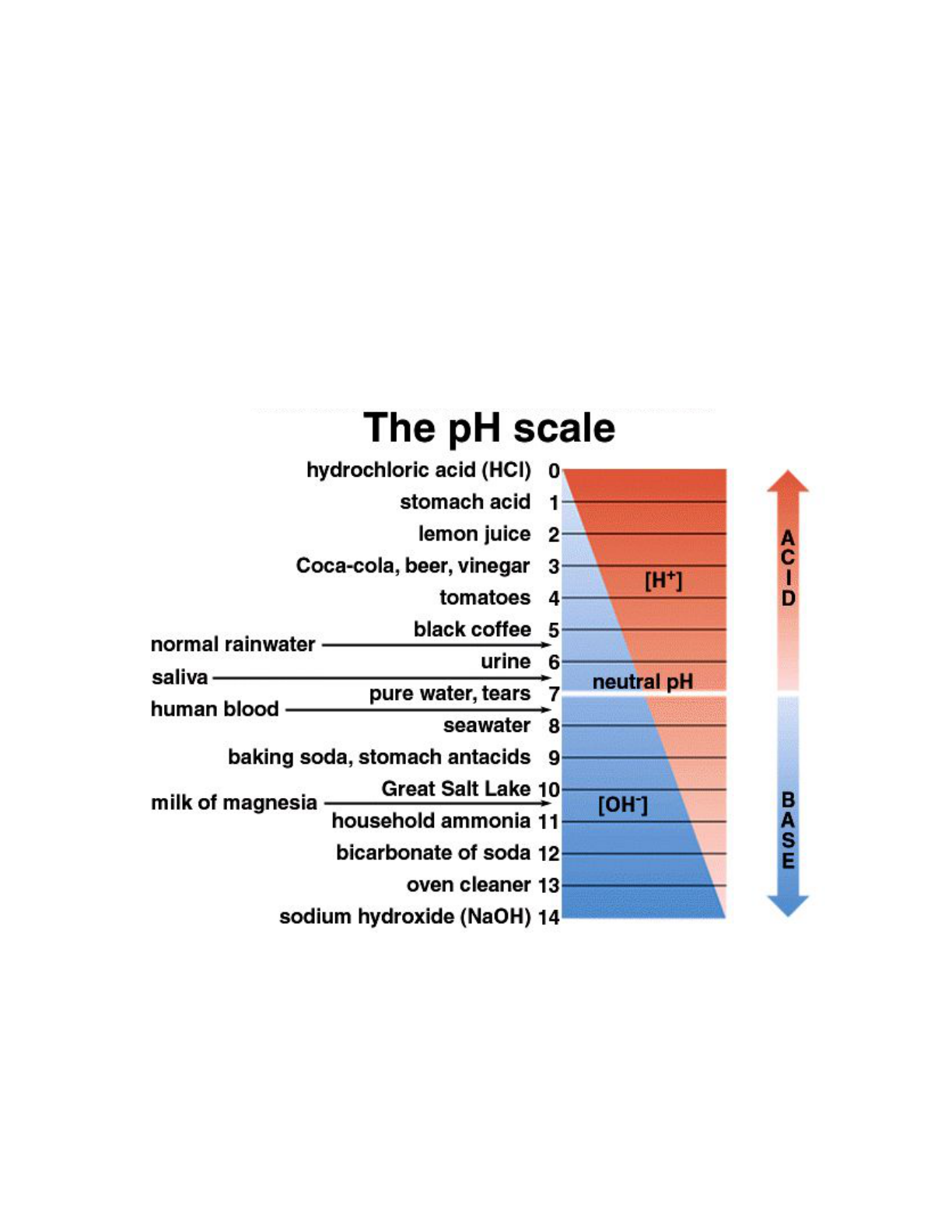
also related to pH. The pH scale goes from 1 to 14 and is the way we measure how acidic a solution is, or
how many hydrogen ions are in solution. Pure water has a pH of 7, which is neutral, which indicates that
it has exactly the same number of H+ (hydrogen) ions as OH- (hydroxide) ions floating in solution.
Two things to remember:
1. If there are relatively more H+ ions, the pH decreases, increasing the acidity. More H+, more
acidic, lower pH.
2. If there are relatively less H+ ions, the pH increases, increasing the basicity. Less H+, more basic,
higher pH.
A pH scale is shown in Figure 3.8. Remember that the pH scale runs from 0 to 14, and each point
represents a tenfold difference because the pH scale is a negative logarithmic scale. Because the scale is
negative, as the ion concentration increases, the pH value decreases. In other words, the more acidic the
solution is, the lower the pH value. A solution with a pH of 5 is 100 times more acidic than something
with a pH of 7. And a solution with a pH of 3 is 10,000 times more acidic than something with a pH of 7.
Figure 3.8 pH Scale. The pH scale ranges from 0 to 14, with 7 being the point of neutrality. What is the pH of lemon
juice? Of milk?
A buffer is a substance that helps to moderate any changes in pH that result from the addition of
acids or bases. This is important because, as you’ll learn later, most of the chemical processes that occur
in living organisms are highly sensitive to pH, and drastic changes in pH can cause serious trouble.
Water can act like a buffer if there is a sudden change in pH. At any given moment, there are a few H
2
O
molecules that break apart and form H+ and OH-. There are a few hydrogen ions here and there who
Chapter 3: Unique Properties of Water 65

effectively get tired of "sharing" an electron with oxygen atoms. These few dissociated water molecules
are what give water its buffering ability. If we add an acid to solution, some of the free OH- ions will bind
to the newly added H+ ions, which will moderate the decrease in pH. Similarly, if we add a base to the
solution, some of the added base will bind to the free H+ ions in solution, which will moderate the
increase in pH. Although water can be a buffer, it isn’t a fantastic one as most of the H
2
O molecules
remain stuck together. It has a little bit of buffering capability and is helpful with small changes in pH,
but it is by no means the best and certainly can’t compensate for super drastic changes in pH.
Acids and Bases
Water is the main ingredient of many solutions. A solution is a mixture of two or more
substances that has the same composition throughout. Some solutions are acids and some are bases.
Recall, in pure water (such as distilled water), a tiny fraction of water molecules naturally breaks down
to form ions. The products of this reaction are a hydronium ion (H
3
O+) and a hydroxide ion (OH-). The
hydroxide ion, which has a negative charge, forms when a water molecule gives up a positively charged
hydrogen ion (H+). The hydronium ion, which has positive charge, forms when another water molecule
accepts the hydrogen ion.
Acidity and pH
Acidity refers to the hydronium ion concentration of a solution. It is measured by pH. In pure
water, the hydronium ion concentration is very low. Only about one in ten million water molecules
naturally dissociates to form a hydronium ion in pure water. This gives water a pH of 7. The hydronium
ions in pure water are also balanced by hydroxide ions, so pure water is neutral (neither an acid nor a
base). Because pure water is neutral, any other solution with the same hydronium ion concentration and
pH is also considered to be neutral.
Acids
If a solution has a higher concentration of hydronium ions and lower pH than pure water, it is
called an acid and has a pH lower than 7. As the hydronium ion concentration increases, the pH value
decreases. Therefore, the more acidic a solution is, the lower its pH value is. Did you ever taste vinegar?
Like other acids, it tastes sour and may sting or burn the skin. Stronger acids can be harmful to
organisms. For example, stomach acid would eat through the stomach if it were not lined with a layer of
mucus. Strong acids can also damage materials, even hard materials such as glass. Testing solutions with
litmus paper is an easy way to identify acids.
Bases
If a solution has a lower concentration of hydronium ions and higher pH than pure water, it is
called a base and has a pH higher than 7. Bases, such as baking soda, have a bitter taste and feel slimy to
the touch. Like strong acids, strong bases can harm organisms and damage materials. For example, lye
can burn the skin, and bleach can remove the color from clothing. Bases, like acids, can be identified
with litmus paper. Bases turn red litmus paper blue.
Neutralization
What do you think would happen if you mixed an acid and a base? If you think the acid and base
would “cancel each other out,” you are right. When an acid and base react, they form a neutral solution
of water and a salt (a molecule composed of a positive and negative ion). This type of reaction is called a
neutralization reaction. For example, when the base sodium hydroxide (NaOH) and hydrochloric acid
(HCl) react, they form a neutral solution of water and the salt sodium chloride (NaCl).
The neutralization reaction is represented by the chemical equation:
NaOH + HCl → NaCl + H
2
O.
In this reaction, hydroxide ions (OH-) from the base combine with hydrogen ions (H+) from the acid to
Chapter 3: Unique Properties of Water 66

form water. The other ions in the solution (Na+) and (Cl-) combine to form sodium chloride.
Lesson Summary
• Most of Earth’s water is salt water in the oceans. Less than 3% is freshwater.
• Water molecules are polar, so they form hydrogen bonds. This gives water unique properties, such as
a polarity, solvency, cohesion, adhesion, high specific heat, and the ability to be a buffering agent.
• A solution is a homogeneous mixture in which a solute dissolves in a solvent. Water is a very common
solvent, especially in organisms. Water is known as the universal solvent.
•Water has high surface tension because of extensive hydrogen bonding.
•The capillary action property of water action is important in moving water upwards through small
spaces. Plants depend on capillary action to move water upward from the roots to the leaves. In the
soil, capillary action also tends to move water upward between the soil particles. Water is moved
through small blood vessels in animals through capillary action also.
•The ion concentration of neutral, pure water gives water a pH of 7 and sets the standard for defining
acids and bases. Acids have a pH lower than 7, and bases have a pH higher than 7.
• Water is involved in most biochemical reactions; such as, photosynthesis and cellular respiration.
Therefore, water is essential to life.
Chapter 3: Unique Properties of Water 67
